Class XII Chemistry: Electrochemistry
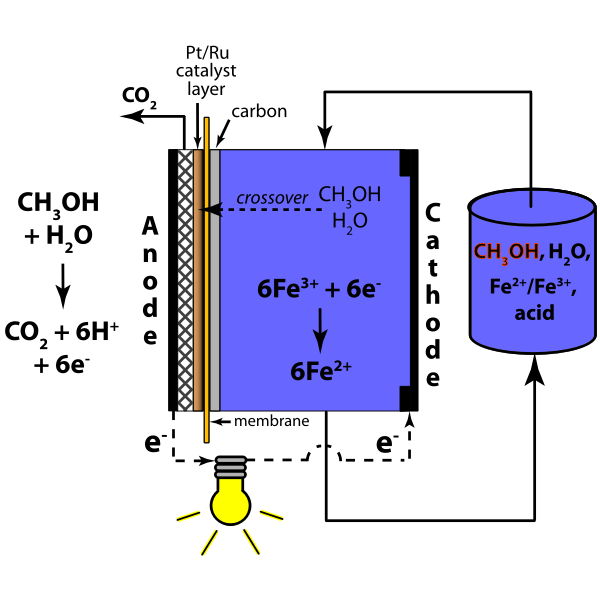
Electrochemistry is a vital branch of chemistry that examines the relationship between electrical energy and chemical change. For Class 12, the focus is on how chemical reactions can produce electricity and how electricity can drive non-spontaneous chemical reactions.
1. Electrochemical Cells
These devices convert energy between chemical and electrical forms. There are two primary types:
- Galvanic (Voltaic) Cells: Convert chemical energy from spontaneous redox reactions into electrical energy (e.g., Daniel Cell).
- Electrolytic Cells: Use electrical energy to drive non-spontaneous chemical reactions (e.g., electrolysis of water).
Standard Electrode Potential (E°)
The potential difference developed between the electrode and the electrolyte is called electrode potential. Under standard conditions (1M concentration, 298K, 1 bar pressure), it is called Standard Electrode Potential.
2. The Nernst Equation
This is the most important quantitative tool in the chapter. It relates the cell potential to the concentration of the species involved.
Ecell = E°cell – (RT/nF) ln ([C]c[D]d / [A]a[B]b)
At 298 K:
Ecell = E°cell – (0.059/n) log Q
3. Conductance of Electrolytic Solutions
Electrolytes conduct electricity through the movement of ions. Key terms include:
- Resistance (R) and Conductance (G): G = 1/R
- Conductivity (κ – Kappa): The conductance of a solution of 1 cm length and 1 sq. cm cross-section area.
- Molar Conductivity (Λm): The conducting power of all ions produced by dissolving one mole of electrolyte in solution.
Kohlrausch’s Law
States that the limiting molar conductivity of an electrolyte can be represented as the sum of the individual contributions of the anion and cation.
4. Electrolysis and Faraday’s Laws
Electrolysis is the process of decomposition of an electrolyte by passing electricity.
- Faraday’s First Law: m = ZIt (where Z is the electrochemical equivalent).
- Faraday’s Second Law: Masses of substances liberated are proportional to their chemical equivalent weights.
5. Batteries and Corrosion
- Primary Batteries: Non-rechargeable (e.g., Dry cell, Mercury cell).
- Secondary Batteries: Rechargeable (e.g., Lead storage battery).
- Fuel Cells: Convert combustion energy of fuels like H2 directly into electricity.
- Corrosion: Electrochemical oxidation of metals (e.g., Rusting: Fe2O3 · xH2O).
Key Formulas Summary
| Concept | Formula |
|---|---|
| Cell Potential | Ecell = Ecathode – Eanode |
| Gibbs Free Energy | ΔG° = -nFE°cell |
| Equilibrium Constant | log Kc = (nE°cell) / 0.059 |
| Cell Constant (G*) | G* = l/A = Rκ |
Advanced Assessment: 20 Thought-Provoking Questions
1. Under what conditions can the Gibbs free energy (ΔG) of a cell reaction be zero, even if the standard cell potential (E°cell) is a non-zero positive value? Discuss the physical significance of this state in terms of battery life.
2. Explain why the molar conductivity of a weak electrolyte like acetic acid increases steeply upon dilution, whereas for a strong electrolyte like KCl, the increase is nearly linear. How does Kohlrausch’s law help in determining Λ°m for weak electrolytes?
3. Hydrogen fuel cells are often cited as the “cleanest” energy source. Analyze the electrochemical challenges regarding the efficiency of oxygen reduction at the cathode. Why is a catalyst like Platinum essential, and what are the alternatives?
4. A Galvanic cell is constructed using two half-cells of the same metal but with different concentrations of their ions. Derive the expression for the cell potential and predict the direction of electron flow. What happens when the concentrations become equal?
5. Compare and contrast the charging and discharging mechanisms of a Lead Storage Battery. Specifically, explain why the density of the electrolyte (H2SO4) changes during these processes and how it serves as an indicator of the battery’s state.
6. In the electrolysis of aqueous NaCl, why is H2 gas evolved at the cathode instead of Sodium metal, but at the anode, Cl2 gas is evolved instead of O2? Discuss the concept of “Overpotential.”
7. How does the temperature coefficient of cell EMF (dE/dT) relate to the entropy change (ΔS) of a chemical reaction? If a cell’s EMF increases with temperature, what can you conclude about the reaction’s spontaneity?
8. Theoretically, can we store Copper Sulphate solution in a Silver vessel? Justify your answer using the Electrochemical Series and explain the role of kinetic versus thermodynamic feasibility in such cases.
9. The conductivity of an electrolyte decreases with dilution, but its molar conductivity increases. Resolve this apparent paradox by explaining the roles of ion concentration per unit volume versus total mobility.
10. Predict the products of electrolysis for a solution of CuSO4 using: (a) Platinum electrodes and (b) Copper electrodes. Explain the electrochemical reasoning behind the difference in products at the anode.
11. Rusting is described as an “electrochemical phenomenon.” Identify the cathode, anode, and electrolyte in a drop of water on an iron surface. Why does the presence of saline water accelerate the process of corrosion?
12. A cell reaction has a very high positive E°cell, yet in practice, the cell provides very little current. What are the internal factors (like internal resistance and polarization) that limit the practical power output?
13. Discuss the validity of Faraday’s Laws of Electrolysis in non-aqueous solvents or molten salts. Are there any conditions where the law of chemical equivalence might appear to fail during electrolysis?
14. If the Standard Hydrogen Electrode (SHE) is assigned a potential of 0.5V instead of 0.0V, how would it affect: (a) E°cell of a Daniel cell and (b) the individual standard reduction potentials of other electrodes?
15. Why is it impossible to measure the absolute potential of a single electrode? Explain the necessity of a reference electrode and the structural requirements for a Salt Bridge to function effectively.
16. Analyze the effect of pH on the potential of the MnO4– / Mn2+ electrode system. How does the Nernst equation predict the oxidizing power of Potassium Permanganate in acidic vs. neutral media?
17. Explain the mechanism of “Cathodic Protection” (Sacrificial Anode) used to prevent the corrosion of underground pipelines. Why is Magnesium or Zinc preferred over Copper for this purpose?
18. For a redox reaction Mn+ + ne– → M, how does the graph of Ered vs. log[Mn+] look? What is the physical significance of the slope and the intercept of this graph?
19. Can the equilibrium constant (Kc) of a reaction be determined if the E°cell is negative? Discuss the relationship between thermodynamic stability and electrochemical potential.
20. In the context of modern green energy, evaluate the role of Lithium-ion batteries. What specific electrochemical properties of Lithium (atomic mass and reduction potential) make it superior to Lead or Nickel?
Industrial & Commercial Electrochemistry: Application-Based Questions
Focus: Engineering challenges, commercial viability, and industrial process optimization.
1. Logistics & Cold Storage: In industrial Lead-Acid battery banks used for backup in cold storage facilities, the capacity significantly drops in winter. As a consultant, explain the electrochemical relationship between temperature and electrolyte viscosity, and suggest how “Internal Resistance” affects the commercial efficiency of these units.
2. Aluminum Industry Economics: The Hall-Héroult process for Aluminum extraction requires massive amounts of electricity. Calculate the commercial impact if the current efficiency drops from 95% to 85% in a plant producing 1000 tons daily. How does “Voltage Drop” across the carbon anodes affect the profit margin?
3. EV Market Strategy: Tesla and other EV manufacturers are moving toward LFP (Lithium Iron Phosphate) batteries over NMC (Nickel Manganese Cobalt). Compare these from a commercial standpoint regarding “Cycle Life” and “Thermal Runaway” risks using electrochemical principles.
4. Industrial Electroplating: A jewelry manufacturer notices that the gold plating on a batch of watches is “pitting” and uneven. Analyze how “Current Density” and the concentration of the complex ion in the electrolyte bath must be managed to ensure a high-quality commercial finish.
5. Maritime Engineering: Shipping companies use “Impressed Current Cathodic Protection” (ICCP) instead of simple sacrificial anodes for large vessel hulls. Explain the commercial advantage of ICCP in terms of maintenance costs and long-term metal preservation.
6. Chlor-Alkali Profitability: In the commercial production of Cl2 and NaOH, the membrane cell has replaced the mercury cell. Discuss the electrochemical and environmental reasons for this shift and how “Brine Purity” affects the life of the ion-exchange membrane.
7. Smartphone Battery Analytics: Smartphone “Fast Charging” technology increases the current significantly. Explain the electrochemical risks of “Lithium Plating” on the anode during high-current charging and how it affects the long-term resale value of the device.
8. Hydrogen Economy: Green Hydrogen is produced via PEM (Proton Exchange Membrane) electrolysis. Analyze the cost-benefit ratio of using Iridium as a catalyst. Why is the “Overpotential” for water splitting the biggest hurdle in making Green Hydrogen price-competitive with fossil fuels?
9. Pipeline Integrity: For a 500km trans-national oil pipeline, calculate the necessity of “Galvanic Anodes” per kilometer based on soil resistivity. How does a change in soil pH from 7 to 5 affect the commercial lifespan of the pipeline?
10. Solar Grid Storage: Redox Flow Batteries (RFBs) are being used for large-scale grid storage. Explain why the commercial scalability of RFBs is superior to traditional solid-state batteries when it comes to decoupling “Power” from “Energy Capacity.”
11. Mining & Electrowinning: In the commercial extraction of Copper from low-grade ores, “Electrowinning” is used. How does the presence of Iron impurities in the leachate solution create a “Current Parasite” effect, reducing the plant’s overall efficiency?
12. Aerospace Battery Tech: Silver-Zinc batteries are used in space missions despite high costs. Analyze the “Specific Energy” (Wh/kg) advantage of this system and explain why the high cost is commercially acceptable in aerospace but not in the consumer car market.
13. Desalination Technology: Electrodialysis (ED) is used to treat brackish water for industrial use. Explain how the “Limiting Current Density” determines the maximum speed of water purification and the commercial risks of “Membrane Fouling.”
14. Recycling Economics: With the rise of EVs, “Urban Mining” of Lithium-ion batteries is a growing business. Discuss the electrochemical challenges of recovering Cobalt vs. Lithium from a spent battery in terms of “Reduction Potential” and energy consumption.
15. Fuel Cell Vehicles (FCVs): For commercial trucking, why is the Hydrogen Fuel Cell considered more viable than heavy Battery Electric systems? Discuss this in terms of “Refueling Time” and “Energy Density” compared to the weight-to-payload ratio.
16. Electronic Waste Prevention: Gold-plated connectors in high-end servers are essential for reliability. Explain the electrochemical advantage of using Gold over Copper in high-humidity data centers to prevent “Contact Resistance” growth over time.
17. Wastewater Treatment: “Electrocoagulation” is used to remove heavy metals from industrial effluent. Compare the commercial efficiency of using Aluminum vs. Iron electrodes in terms of “Sludge Production” and power consumption.
18. Smart Grid Integration: “Vehicle-to-Grid” (V2G) technology allows EVs to sell power back to the grid. Analyze how the frequent “Micro-Cycling” of the battery affects its electrochemical health and the commercial insurance warranties provided by car manufacturers.
19. Sensors & Medical Tech: Commercial glucose monitors use electrochemical biosensors. Explain how the “Amperometric” detection of glucose works and why “Selectivity” against other blood components is the most critical commercial feature of the sensor strip.
20. Submarine Power Systems: Modern non-nuclear submarines use Stirling engines or Fuel Cells (AIP – Air Independent Propulsion). Discuss the electrochemical signature and “Silent Operation” benefits of Fuel Cells over traditional Diesel-Electric generators in naval defense commerce.


